During ZEN DNA extractions we noticed that samples preserved in the Zymo buffer were forming a precipitate with the C1 solution from the MoBio kit. Furthermore, many of the samples also resulted in very low DNA yields, perhaps correlated with the precipitate formation. Any microbiologist will tell you that there are many different ways to preserve samples from the field, but there does not seem to be a universal *best* method.
We decided the best way to approach this problem for the Seagrass Microbiome project was to explore a variety of sample preservation methods and see which approximated the ‘real’ microbiome best (as measured by preservation on dry ice). We chose the following methods to try: Dry ice, Zymo, RNA-later, Drierite, and Ethanol. We also wanted to investigate how different preservation methods performed over time. We chose 4 time points post sampling for our extractions: 24 hours, 1 week, 2 weeks, and 1 month.
We drove to Putah Creek in Winters in search of submerged aquatic plants in the Alismatales (the order that contains the seagrasses). Here is a photo of our study site: 
We found a bed of Elodea canadensis growing near the shore and started sampling.
We pulled out whole plants and divided them into root and leaf for each of our trial preservation methods, with 3 replicates per method per time point. We extracted DNA at each of the different time points, and did PCR of the 16S SSU rRNA gene, with subsequent library preparation and sequencing on the Illumina Mi-Seq. We used QIIME to analyze the sequence data and compare species diversity between our preservation methods.
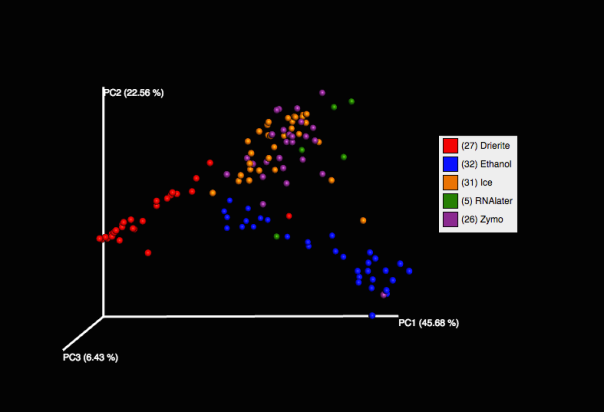
Our PCoA plot shows that preservation in Zymo best approximates the community captured using dry ice (our control), and that Drierite and Ethanol were both very different from anything else. RNA-later appears close to Zymo and dry ice, but we only have 5 data points for it. For the RNA-later and Ethanol extractions, we extracted directly from the solution without pelleting, which may have affected our end result.
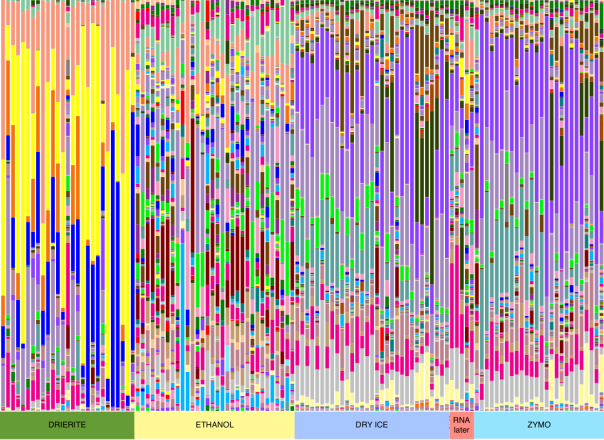
The taxonomy table supports the trend seen in our PCoA plot, with Drierite and Ethanol being very different, and Zymo, dry ice, and our 5 RNA-later time points showing similar communities.
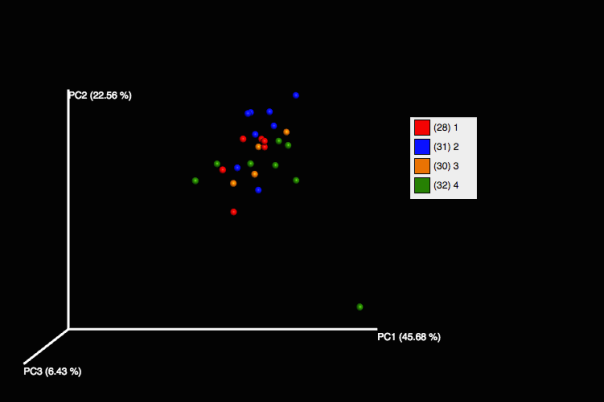
Looking at variation in community structure across time points within Zymo we see little to no change.
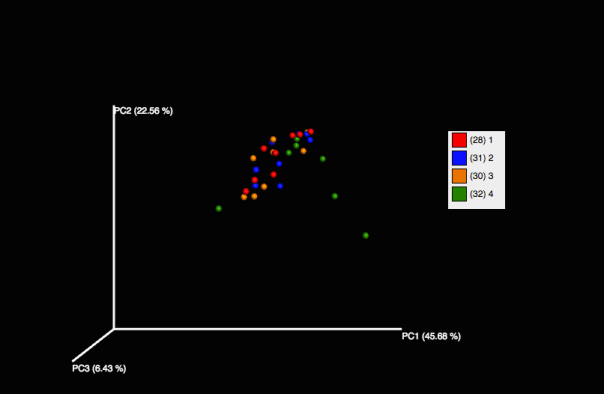
However, we see some change in community structure by the month mark with dry ice.
Take home messages from our sample preservation experiment:
- Using Drierite as a preservation technique does not capture the community assembly well.
- RNA-later and Ethanol could have had better success with pelleting and removing the supernatant prior to extraction. We may investigate this further in the future.
- In spite of our initial concerns regarding precipitate forming in the Zymo buffer, Zymo is the clear winner in our trial.
- There are slight changes in the community assembly by the month mark, in all methods (depending on whether you use weighted or unweighted unifrac PCoA).
- Due to the current ease of sampling using dry ice, we will continue this method except in situations where samples must be kept at room temperature (we will then use Zymo).
HAPPY SAMPLING 🙂 Please see https://seagrassmicrobiome.org/sample-collection-and-preservation/ for more details regarding sampling and preservation protocols.

